Cervical Spine Center
Kalra Brain & Spine
At KBS, we treat our patients as neighbors, friends, and family. Our goal is to educate you, treat your symptoms, and help you recover as fast as possible.
A second opinion may save you from a cervical fusion.
Cervical Spine Center
Dr. Kalra was one of the first physicians trained in implanting the Mobi-C artificial disc when it became approved by insurance companies. He strongly believes that patients who desire a minimally invasive surgery, no down-time, and no cervical collar, should undergo cervical arthroplasty instead of a fusion. This option preserves the natural motion of the neck and substantially decreases the risk of another neck surgery down the road.
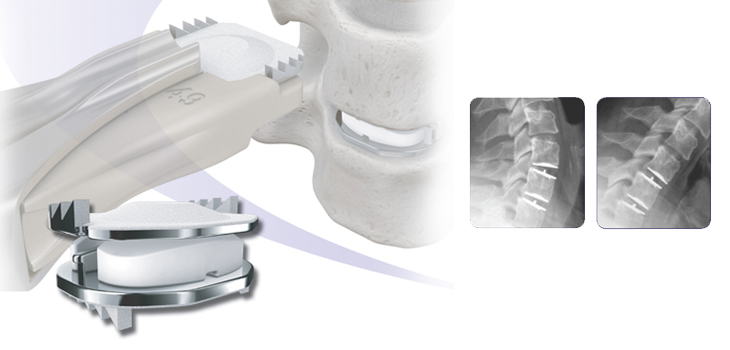
Mobi-C® Cervical Disc
- Only FDA Approved Cervical Disc for One & Two Levels
- Superior to fusion for two-level disc replacement
The Mobi-C cervical disc is one of the most popular artificial discs in the world. It was first implanted in Orleans, France in November 2004. Since then, Mobi-C has been implanted in more than 20,000 spinal levels.
How the Disc Works
Mobi-C contains patented Mobile Core Technology™ that allows the polyethylene core to slide and rotate inside the disc for self-adjustment to the cervical spine movements. This means that Mobi-C can imitate the normal motion of the cervical spine.
Mobi-C provides bone sparing fixation without chisel cuts into the small vertebral bodies of the cervical spine, making it safe for implantation at one or two levels.
Mobi-C Clinical Trial Results
In the one-level and two-level studies, Mobi-C demonstrated reduced rates of adjacent level disease.
- Overall trial success was 69.7% for Mobi-C and 37.4% for fusion.
- The rate of secondary surgeries at the operated level for Mobi-C was 3.1% versus 11.4% for fusion.
- The percentage of subjects who demonstrated adjacent segment degeneration (determined by x-ray) was:
- 2.9% of Mobi-C patients compared to 18.1% of ACDF patients at the inferior adjacent level
- 13.1% of Mobi-C patients compared to 33.3% of ACDF patients at the superior adjacent level
- Mean return to work time was 20.9 days shorter for Mobi-C patients compared to fusion patients.
Request an Appointment
Contact us
Get In Touch
Address
Kalra Brain & Spine
5899 Preston Road
Suite 1303
Frisco, TX 75034
Phone
972-905-9226

Fax
972-905-9269